Dimethyl malonate
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 108-59-8
- EC NO : 203-597-8
- Molecular Formula : C5H8O4
- Main Specifications : 99% min
- Synonyms : dimethyl propanedioate;Methyl malonate;dimethylpropanedioate;Dimethyl ester of malonic acid;
Package: 25kg����,50kg 200kg
Uses : As an organic synthesis intermediate
Molecular Structure:
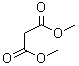
Product description:
What is the chemical of Dimethyl malonate ?
Appearance: Colorless liquid, soluble in organic solvents such as alcohols and ethers, slightly soluble in water ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point:181°C;
===============================================================================
As an organic synthesis intermediate, it is widely used in the synthesis processes of pharmaceuticals, pesticides, fragrances, dyes, and resins, such as being a key raw material for the production of pharmaceutical pipemidic acid; At the same time, it can be used as a solvent, diluent, or reagent in industry, applied in chemical fields such as coatings, inks, plastics, and lithium battery electrolytes.
=============================================================================
In industry, the production of dimethyl malonate mainly adopts the cyanide esterification method, which includes the steps of: neutralizing chloroacetic acid with sodium carbonate to produce sodium chloroacetate, and then reacting with sodium cyanide to produce sodium cyanoacetate; Sodium cyanide is hydrolyzed to obtain sodium malonate, which is then esterified with methanol in the presence of sulfuric acid and purified by washing and distillation to obtain the final product. In addition, the catalytic carbonylation method developed abroad uses chloroacetate, carbon monoxide, and methanol as raw materials, and synthesizes them in the next step under the action of catalysts. However, this method has a complex process and strict reaction conditions, which poses challenges for industrial application
