2-Tridecanone
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 593-08-8
- EC NO : 209-784-0
- Molecular Formula : C13H26O
- Main Specifications : 99% min
- Synonyms : Methyl undecyl ketone;
Package: 25kg 50kg drum or bag
Uses : As a food flavor,
Molecular Structure:
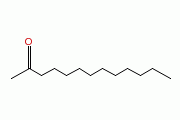
Product description:
What is the chemical of 2-Tridecanone ?
Appearance: White wax or colorless crystalline flakes with wax, fat, dairy, and coconut aromas, and nutty and herbal notes. At room temperature, it can appear as a powder, block or transparent liquid, with a color of white or colorless to light orange to yellow. ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 24–27 °C;
boiling point: 263 ℃ (at 760 mmHg);
========================================================================
Purpose:
As a food flavor, it is allowed to be used in essence such as mushrooms, coconuts, jackfruit, chicken, cheese, smoked pork, etc. according to the provisions of GB 2760-1996, with a limit of 3.0 mg/kg (cold drinks, candy, baked goods, etc.).
Used as a fragrance enhancer (PERFUMING) in cosmetics and personal care products.
In agriculture, 2-tetradecanone isolated from wild tomatoes has non alkaloid insecticide activity and can be used as an insect repellent.
As an organic synthesis intermediate, it is used to prepare aliphatic ketone essence and thirteen carbon chain derivatives
=============================================================================
Synthesis route:
In industry, it can be prepared by reacting a mixture of lauric acid and acetic acid with thorium oxide catalyst at 450 ℃.
The laboratory synthesis method includes: using dodecanoic acid as raw material, reacting with methyl lithium or methyl Grignard reagent after acylation, and then hydrolyzing to obtain the product; Alternatively, the Friedel Crafts acylation reaction can be carried out using undecane and acetyl chloride under the catalysis of aluminum trichloride, with yields ranging from 75% to 85%.
