Sodium Perchlorate
-
- Category :
Inorganic chemicals/Inorganic salts
- CAS NO : 7601-89-0
- EC NO : 231-511-9
- Molecular Formula : NaClO4
- Main Specifications : sodium perchlorate
- Synonyms : Sodium perchlorate hydrate;Sodium Perchlorate, Anhydrous;Sodium Perchlorate Anhydrous;
Package: 25kgs/bag. 1000kgs/bag
Uses : Sodium perchlorate is the precursor to many other perchlorate salts, often taking advantage of their low solubility relative to NaClO4 (209 g/100 mL at 25 °C). Perchloric acid is made by treating NaClO4 with HCl.NaCIO4 finds only minimal use in pyrotechni
Molecular Structure:
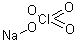
Product description:
Sodium perchlorate is the inorganic compound with the formula NaClO4. It is the most soluble of the common perchlorate salts. It is a white crystalline, hygroscopic solid that is highly soluble in water and in alcohol. It usually comes as the monohydrate, which has a rhombic crystal structure.
Uses
Sodium perchlorate is the precursor to many other perchlorate salts, often taking advantage of their low solubility relative to NaClO4 (209 g/100 mL at 25 °C). Perchloric acid is made by treating NaClO4 with HCl.NaCIO4 finds only minimal use in pyrotechnics because it is hygroscopic; ammonium and potassium perchlorates are preferred. They are made by double decomposition from a solution of sodium perchlorate and potassium or ammonium chlorides.
Laboratory applications
NaClO4 has a variety of uses in the laboratory, often as a nonreactive electrolyte. For example, it is used in standard DNA extraction and hybridization reactions in molecular biology.
Production
Sodium perchlorate is produced by anodic oxidation of sodium chlorate, not sodium chloride, at a platinum electrode.
ClO3 + H2O → ClO4 + 2 H2
